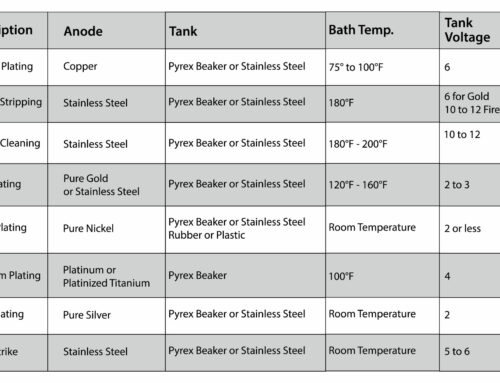
Gold isn’t the only metal you might find yourself in a position to buy. You may find yourself confronted with people wanting to sell platinum. Use this guide to learn the ins and outs of testing platinum. When you are testing for platinum, you will approach the process in much the same way you would with testing gold: you’ll use the testing stone again and use acid to determine the metal content of the piece you are testing.
Tools Needed:
- A powerful magnet
- A testing stone
- And 22 karat gold or platinum testing acid
Step 1
One thing to pay attention to and be aware of before testing for platinum is that iron and steel items will pass the streak test for platinum so before attempting a streak test, you should always start by testing any suspected platinum with a powerful magnet.
If the metal is attracted (strongly) to the magnet, it can be one indicator – but Cobalt is sometimes present in 950 platinum so its not a defining test.
Step 2
Once you have ruled out steel and iron, take the item and scratch it along the surface of the stone like you would with any piece of gold. Make sure that you have a thick and visible deposit line that you can test accurately and is about 1 inch long.
Step 3
Apply a drop of your platinum testing acid across the streak on your testing stone.
Step 4
Identify the reaction. If the metal you are testing is platinum, the streak should keep its bright white coloring for over a minute of exposure to the testing acid.
Be aware that other than iron and steel, there are plenty of other white non-magnetic metals that will give false platinum reading like chromium cobalt, dental materials, some class rings and some metals frequently used in stainless flatware. Always try to verify your results in more than one way whenever possible.


Hi. My wife’s £15,000 platinum engagement ring is magnetic and a neodymium magnet will pick it up easily. This is due to it being alloyed with 5% cobalt, the other 95% is fine platinum.
Your comment ‘If the metal is attracted to the magnet, you are not dealing with platinum’ isn’t really correct in all cases. Platinum is difficult to identify, and wasn’t even hallmarked or assayed in the UK until 1975, so all suspect platinum jewelry should go the full range of tests before dismissing
I have 925 sterling silver ring plated in Rhodium. The acid testing kit I have only comes with acid to test gold, silver, and platinum acid liquid. Would the platinum acid work testing rhodium?
can any one tell me what it means when i put the acid on the platinum why does it smoke
If I have platinum and the acid scratch test confirms it and direct acid on my platinum has no reaction and an xrf exray machine at iPawn says 99.9% platinum then can I be pretty sure it’s platinum?
I have a rock and the silvery shiny material peeking through at the bottom, in tiny spots. I dug out some matter, but mostly dust and flakes, no nuggets to be able to scratch the black stone. Can I put the flakes on the stone and just drop the Platinum drops, direct on the flakes? and I was told that they are not always reliable? How accurate do you think?
I have a display item and done a lot of test, dsnt react to acid test, dsnt melt, dnt attract to magnet, that’s why we suspect it has a platinum content. It dsnt evn discoulor for hw mny yrs and stays it bright color, is it possible that it is a real platinum. A lot of jewelers tried to melt it bt stays as is. Hope you can help me identify its metal content. Thank you.
Does pewter react to platinumacid test? Will the line appear or disappear with the acid scratch test?
I life in Papua New Guinea I found a silver colored and it’s is 30cm long and it’s look like cold bar but is coloured is silver and I think is platinum I put magnets but no magnet apply it just slight so please can you help me with some ideas of test