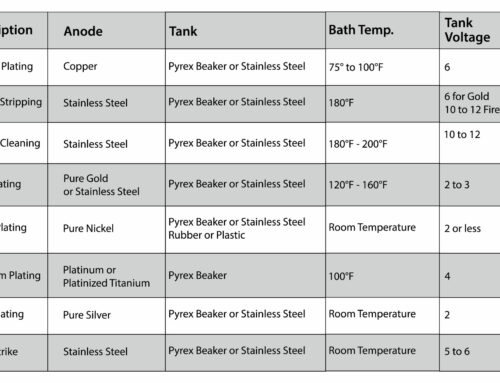
For this method of testing silver that you suspect of being plated or of a different value than your initial tests suggested, you will test it much the same way you test the silver non-destructively with the scratch test. The only difference is that you will cut the metal of the unknown silver piece and open it so you can scratch the metal from the inside of the piece against the testing stone instead of the surface metal. Remember, you should only use this test if you are unsure of the results you got from the basic streak test initially or suspect the metal is not actually silver. And always be sure to check with your customer before performing a destructive test on their valuable metals.
Tools Needed:
- A jewelry cutter
- A jeweler’s pliers
- A testing stone
- An optional jeweler’s loupe or other magnification device
- And silver testing acid
Step 1
Take your jewelry cutters to cut through the metal so you have an opening in the piece of silver. This may take several tries to create an opening if the metal is particularly thick or heavy.
Step 2
Once you have a cut all the way through the metal, take your jewelry pliers and pull the metal apart to open the gap. Continue prying the metal apart until the gap is wide enough or positioned so that you can rub the inside portion of the metal against the testing plate to create a streak like you did before using the outside surface of the metal.
Step 3
Rub the inside section of the metal piece directly against the surface of the testing stone so that it leaves a mark on the testing stone. Press firmly on the testing stone to leave a thick and visible deposit that you can test accurately. The mark should be about 1 to 1 1/2 inches long.
Step 4
Place a drop or two of the silver testing acid onto the scratch mark you made with the inside metal of the suspected silver piece. Observe the reaction. You may find that using a jeweler’s loupe or other magnification device will make it easier to see the small changes in acid color to read the test results accurately.
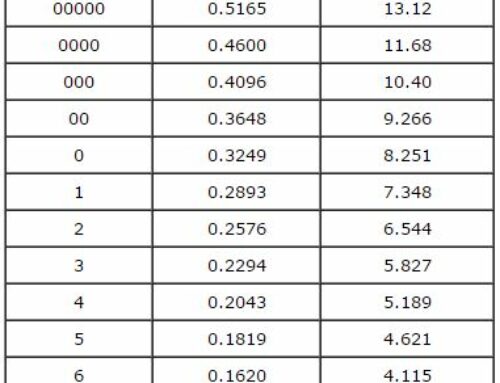
- If the item has a silver content of 90 to 100%, the acid will turn a light creamy color. The lighter the cream color is, the higher the silver content.
- If the item has a silver content between 77 and 90%, the acid will turn a gray color.
- If the item has a silver content between 65 and 75%, then the acid will turn a light green color.
- And if the acid turns very strongly green immediately with profuse bubbles, the item is not solid silver.


Leave A Comment